Biopharmaceuticals can exhibit tremendous heterogeneity in terms of posttranslational modifications (PTMs) such as glycosylation, oxidation, deamidation, phosphorylation etc. and structural modifications. In addition to this, changes in upstream and downstream processes during recombinant production of biologics also influence structural heterogeneity. Therefore, comprehensive characterization of biologics is necessary to ensure safety and efficacy.

We are working towards making the Biologics Characterization Facility compliant to ISO standards.
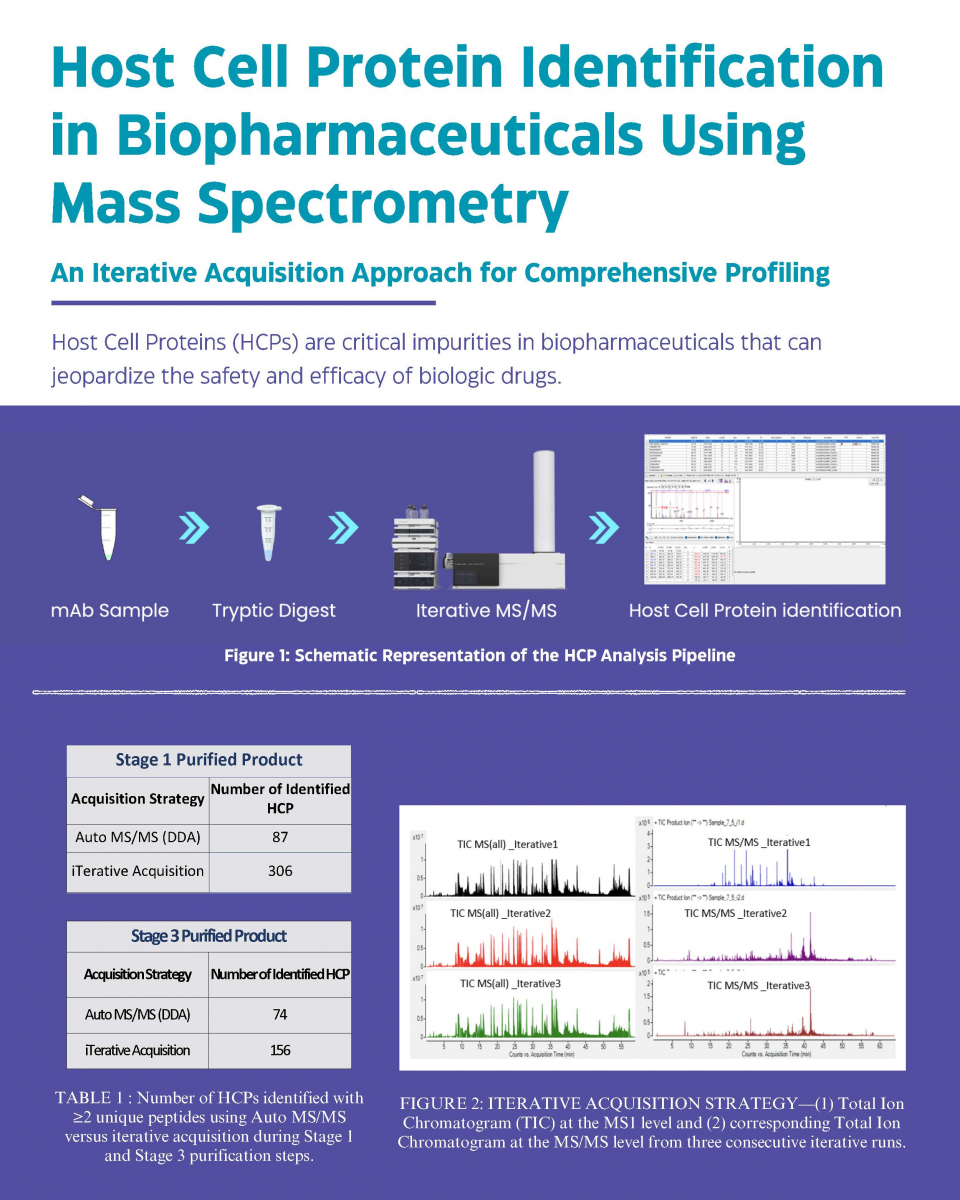
Case Study 1 - Host Cell Protein ID in Biopharmaceuticals using Mass Spectrometry
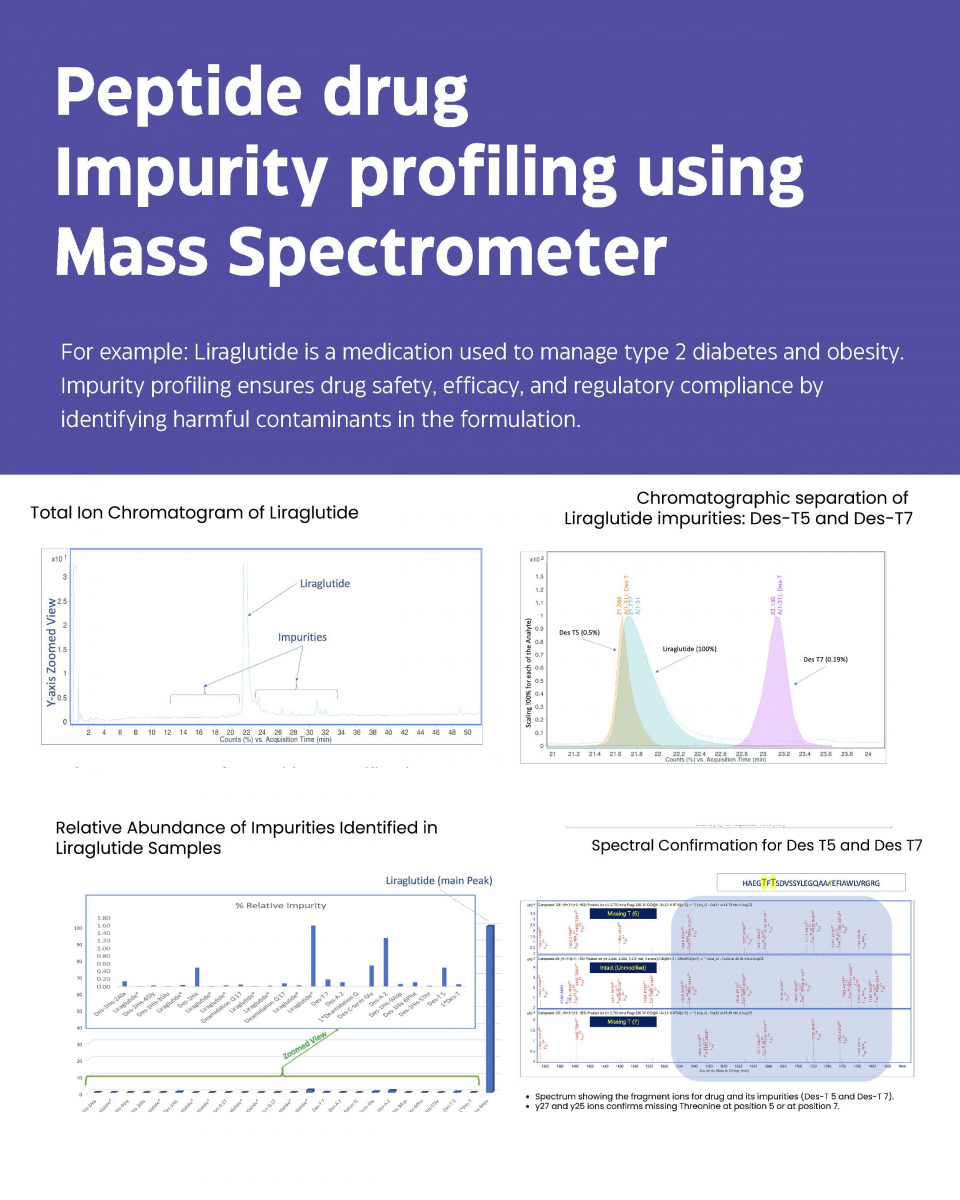
Case Study 2 - Impurity Profiling in Peptide drugs by Mass Spectrometry